China’s National Medical Products Administration (NMPA) has given a conditional approval for the public use of its second locally made COVID-19 vaccine — CoronaVac. The approval comes a month after
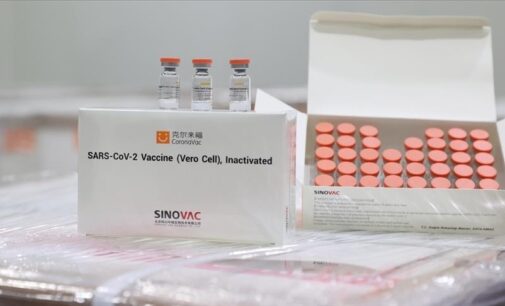
China’s National Medical Products Administration (NMPA) has given a conditional approval for the public use of its second locally made COVID-19 vaccine — CoronaVac. The approval comes a month after